Clean-in-place (CIP) systems are automated cleaning systems that flush cleaning solutions through equipment without requiring disassembly. In meat packaging environments this matters because product residues are rich in protein and fat—conditions that allow bacteria to multiply quickly if sanitation is inconsistent. Food plants must thoroughly clean equipment between production runs while minimizing downtime. CIP systems solve this by automating the cleaning process across production lines.
This guide explains how clean in place food processing systems work, how they compare to manual cleaning, and how they support CIP food safety and regulatory compliance in modern meat packaging facilities.
What Does CIP Stand For?
CIP stands for Clean-in-Place. It refers to an automated sanitation method that cleans the internal surfaces of equipment—pipes, tanks, fillers, conveyors, and packaging machines—by circulating cleaning solutions through the system at controlled temperatures and flow rates. The equipment remains assembled during the process; operators do not dismantle the line to clean it.
A programmed sequence typically runs several stages: rinse, wash, rinse, and sanitize. Each stage uses specific temperatures, chemical concentrations, and circulation times to remove food residues and microorganisms.
CIP is often paired with COP (Clean-out-of-Place). COP applies to removable parts that are taken off the line and cleaned separately in wash stations. In meat processing plants the two systems work together: CIP handles fixed equipment while COP addresses removable components such as guards, trays, and detachable fittings.
How CIP Works — Step by Step
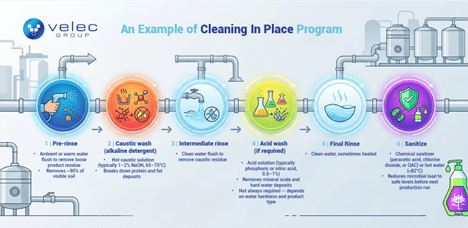
Below is a typical CIP cycle used for automatic cleaning of food production lines in meat processing.
Step 1: Pre-rinse
→ Ambient or warm water flush to remove loose product residue
→ Duration: 3–5 minutes
→ Removes ~90% of visible soil
Step 2: Caustic wash (alkaline detergent)
→ Hot caustic solution (typically 1–2% NaOH, 65–75°C)
→ Duration: 10–20 minutes
→ Breaks down protein and fat deposits
Step 3: Intermediate rinse
→ Clean water flush to remove caustic residue
→ Duration: 3–5 minutes
Step 4: Acid wash (if required)
→ Acid solution (typically phosphoric or nitric acid, 0.5–1%)
→ Duration: 5–10 minutes
→ Removes mineral scale and hard water deposits
→ Not always required — depends on water hardness and product type
Step 5: Final rinse
→ Clean water, sometimes heated
→ Duration: 3–5 minutes
Step 6: Sanitize
→ Chemical sanitizer (peracetic acid, chlorine dioxide, or QAC) or hot water (>82°C)
→ Duration: 5–10 minutes
→ Reduces microbial load to safe levels before next production run
A full CIP cycle for a typical meat packaging line runs 1h45 minutes, depending on equipment complexity and soil levels. Modern systems store multiple cleaning “recipes,” allowing different machines or production zones to run customized cycles. Sensors monitor flow rate, temperature, chemical concentration, and conductivity throughout the cycle. Each parameter is logged automatically, producing verification records that can be reviewed during food safety audits.
CIP vs Manual Cleaning
In many meat processing plants the decision between CIP and manual cleaning is primarily an operational question. Manual sanitation relies on operators to dismantle equipment, spray surfaces, scrub residues, and reassemble the line. CIP systems automate most of that work.
Factor | CIP (Automated) | Manual Cleaning |
|---|---|---|
Cleaning time per cycle | 45–90 minutes | 2–4 hours |
Labor required | 1 operator (monitoring) | 3–6 operators (active cleaning) |
Water usage per cycle | 200–500 liters (recirculated) | 500–1,500 liters (single-pass) |
Chemical usage | Precise, metered dosing | Variable — operator-dependent |
Cleaning consistency | Repeatable — same parameters every cycle | Variable — depends on operator skill and diligence |
Documentation | Automatic data logging (time, temp, concentration, flow) | Manual log sheets — often incomplete |
Equipment disassembly required | Minimal to none | Significant — guards, conveyors, nozzles |
Validation / audit readiness | High — automated records satisfy BRC, SQF, FSSC 22000 | Low — difficult to prove consistency |
Risk of recontamination | Low — closed system | Higher — open equipment, manual handling |
Capital cost | Higher (CIP station, piping, controls) | Lower (hoses, spray guns, tanks) |
Operating cost per cycle | Lower (less water, chemical, labor) | Higher (more labor, water, chemical waste) |
Typical payback period | 12–24 months at production volumes >40 hours/week | — |
The financial impact is usually straightforward to calculate. A line operating two shifts per day and sanitized daily can recover 1.5–3 hours of production time per cleaning event when switching from manual cleaning to CIP. Across a full year that represents roughly 400–750 hours of additional production capacity. At even modest output values, the recovered production time alone often offsets the capital investment in a CIP system within 12–24 months.
Regulatory Requirements
Food safety regulations rarely mandate specific sanitation technologies, but they do require facilities to demonstrate effective and documented cleaning procedures. CIP systems align closely with those expectations.
FDA / FSMA (United States) Under the Food Safety Modernization Act, food facilities must implement preventive controls that include monitored sanitation procedures. Facilities must verify that cleaning occurs as planned and maintain records demonstrating compliance. CIP systems automatically log parameters such as temperature, time, and chemical concentration, creating documentation that satisfies these monitoring and verification requirements.
EU Regulation 852/2004 This regulation requires food business operators to maintain equipment that can be cleaned effectively and, when necessary, disinfected. It also requires equipment design that allows adequate sanitation. While CIP is not explicitly required, automated cleaning systems support the regulation’s emphasis on hygienic design and documented cleaning processes.
Third-party food safety certification programs (BRC, SQF, FSSC 22000) Most large food manufacturers operate under certification schemes that audit sanitation programs closely. Auditors typically review cleaning procedures, parameter monitoring, and sanitation records. CIP systems provide automatically recorded data showing each stage of the cleaning cycle, which simplifies verification during audits.
For meat packaging operations in particular, sanitation programs focus heavily on pathogens such as Listeria monocytogenes, Salmonella, and pathogenic E. coli. Automated CIP monitoring improves traceability and consistency in sanitation programs—an advantage when demonstrating compliance with modern CIP food safety expectations.
CIP systems are also one part of a broader hygienic design strategy. Equipment geometry, drainage, and accessibility all affect sanitation effectiveness. (See Velec’s guide to hygienic design for food processing equipment for more on that topic.)
Where CIP Fits in a Meat Packaging Line
CIP systems typically connect to several areas of a packaging line where residues accumulate and sanitation must occur frequently. These include:
- Product transfer piping and pumps
- Filling and dosing systems
- Conveyors and product handling equipment
- Packaging machines and thermoformers
- Intermediate storage tanks or buffers
In sausage packaging lines, for example, product residues can accumulate in transfer pipes and filling systems during production. Integrating CIP allows operators to flush these areas quickly between batches. (See the complete sausage packaging line solution for an example of a CIP-ready configuration.)
Conveyors are another frequent sanitation point in automated packaging environments. Equipment designed for CIP compatibility typically includes self-draining frames, smooth welds, and accessible spray coverage. Velec’s conveyor solutions incorporate hygienic features that support both manual cleaning and CIP integration.
Sanitation performance also depends on broader equipment design choices. Automated cleaning systems work best when paired with hygienic surfaces, minimal dead zones, and tool-free access points. (See the article on ensuring food safety in automated processing lines for more context.)
FAQ
What does CIP stand for in food safety?
CIP stands for Clean-in-Place. It is an automated sanitation method used in food processing that circulates cleaning solutions through equipment without requiring disassembly. The system runs programmed cleaning cycles using controlled temperatures, flow rates, and chemical concentrations to remove food residues and microorganisms between production runs.
What is the difference between CIP and manual cleaning?
CIP systems clean equipment automatically by circulating cleaning solutions through the line. Parameters such as temperature, chemical concentration, and flow rate are programmed and monitored for consistency. Manual cleaning relies on operators to dismantle equipment and clean surfaces with hoses and scrubbing tools. CIP cycles are faster, more consistent, and produce automatic sanitation records, while manual cleaning is more labor-intensive and harder to document.
Is CIP required by food safety regulations?
Most food safety regulations do not specifically require CIP systems. However, frameworks such as FSMA in the United States, EU Regulation 852/2004, and third-party audit standards like BRC, SQF, and FSSC 22000 require documented sanitation procedures and verified cleaning effectiveness. CIP systems simplify compliance by automatically recording the parameters of each cleaning cycle.
How long does a CIP cycle take?
A typical CIP cycle in a meat packaging line takes 1h45 minutes. The process includes a pre-rinse, caustic wash, intermediate rinse, optional acid wash, final rinse, and sanitization stage. The exact duration depends on equipment complexity and the amount of residue present. Equivalent manual cleaning processes usually require 2–4 hours without the same cleaning garantees.
Clean-in-Place Systems and Hygienic Packaging Equipment
Clean-in-place systems work best when equipment is designed to support sanitation from the start. Surfaces must drain properly, crevices must be minimized, and connections must allow cleaning solution flow throughout the system.
Velec packaging lines incorporate these principles across our equipment range, including CIP-compatible connections, hygienic conveyor designs, and tool-free disassembly where manual access is required.
Velec packaging lines are designed with CIP integration from the outset — tool-free disassembly, self-draining surfaces, and CIP-compatible connections are standard across our equipment range.
Talk to our engineering team about CIP-ready line configurations →







